Immunotherapy for Bladder Cancer
BCG W/ INTERFERON - Used if BCG fails. A review of 5 RCT with 1231 patients showed low-quality evidence suggesting BCG alternating with IFN-α compared with BCG alone may increase time-to-recurrence, however low-quality evidence also suggests no clear differences for time-to-progression or discontinuation of therapy due to adverse events. This study observed no clear difference between BCG plus IFN-α vs. BCG alone for recurrence (average risk ratio (RR) 0.76, 95% confidence interval (CI) 0.44 to 1.32; 4 RCTs; 925 participants; very low-quality evidence) or progression (average RR 0.26, 95% CI 0.04 to 1.87; 2 RCTs; 219 participants; low-quality evidence). The subject of BCG + interferon lacks high quality, well powered and well designed trials (3)
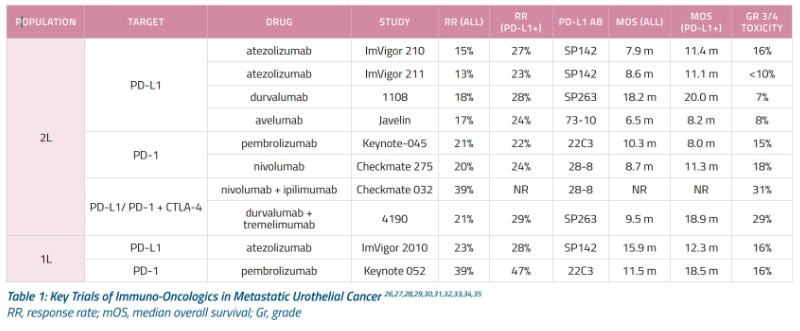
ANTI-PD1 = Pemborlizumab, Nivolumab
ANTI-PDL1= Atezolizumab, Durvulumab, Avelumab
PEMBROLIZUMAB - Approved for 1st line therapy for platinum ineligible mUC patients. A study published in 2021 looked into neoadjuvant pembrolizumab with encouraging results. (12) Please click here for more information
NIVOLUMAB - Anti-PD1 FDA approved for platinum refractory m UC. Phase II CheckMate 275 evaluated monotherapy in mUC or unresectable platinum resistant UC. A 2021 NEJM study reported improved outcomes with adjuvant nivolumab in high risk patients. (13)
ATEZOLIZUMAB - anti-PDL-1 monoclonal antibody approved for first line therapy in mUC platinum ineligible patients. Approved based on IMvigor210 study of platinum refractory mUC. IMvigor211 compared to chemotherapy. Provides 2.1mo PFS and 11mo OS in mUCC with 46% 12mo survival rate (7). Used as neoadjuvant, the phase II ABACUS study found a 29% complete response (T0) on final pathology and a 39% downgrading to NMIBC. (9) A 2020 study found that PPI use was associated with worse outcomes when atezolizumab was used. (11) The FDA issued a May 2018 safety alert that people with low PD-L1 expression have decreased survival with pembrolizumab or atezolizumab compared to cisplatin or carboplatin based therapy. In August they added the requirement for a diagnostic test be done in anyone on these drugs to test for PD-L1 expression.(5)
DURVALUMAB - MEDI4736 study in PDL-1i for use in metastatic bladder cancer, showed 46.4% response rate and granted accelerate approval by FDA in May 2017. (2)
AVELUMAB - Received accelerated approval in 2017 based on JAVELIN study.
COMBO THERAPY - CheckMate 032 included nivolumab/ipilimumab
ERDAFITINIB - Post chemotherapy, post immunotherapy, FGF receptor target. NEJM published study in 2019 showed objective tumor response in 40% patients after disease post chemotherapy progression. Provided 5.5mo PFS, 13.8 OS. This study included patients with visceral metastasis and more than half w/ GFR <60. (6)
ENFORTUMAB
SIDE EFFECTS OF IMMUNOTHERAPY
Immune mediated adverse events (IMAE's) are activively monitored. Any organ system can be affected and neuropathies and arthralgias can also occure. The most commonly experienced one may include pneumoitis., colitis, dermatitis, hepatic transaminitis and thyroiditis. (10)
ANTI-PD1 = Pemborlizumab, Nivolumab
ANTI-PDL1= Atezolizumab, Durvulumab, Avelumab
PEMBROLIZUMAB - Approved for 1st line therapy for platinum ineligible mUC patients. A study published in 2021 looked into neoadjuvant pembrolizumab with encouraging results. (12) Please click here for more information
NIVOLUMAB - Anti-PD1 FDA approved for platinum refractory m UC. Phase II CheckMate 275 evaluated monotherapy in mUC or unresectable platinum resistant UC. A 2021 NEJM study reported improved outcomes with adjuvant nivolumab in high risk patients. (13)
ATEZOLIZUMAB - anti-PDL-1 monoclonal antibody approved for first line therapy in mUC platinum ineligible patients. Approved based on IMvigor210 study of platinum refractory mUC. IMvigor211 compared to chemotherapy. Provides 2.1mo PFS and 11mo OS in mUCC with 46% 12mo survival rate (7). Used as neoadjuvant, the phase II ABACUS study found a 29% complete response (T0) on final pathology and a 39% downgrading to NMIBC. (9) A 2020 study found that PPI use was associated with worse outcomes when atezolizumab was used. (11) The FDA issued a May 2018 safety alert that people with low PD-L1 expression have decreased survival with pembrolizumab or atezolizumab compared to cisplatin or carboplatin based therapy. In August they added the requirement for a diagnostic test be done in anyone on these drugs to test for PD-L1 expression.(5)
DURVALUMAB - MEDI4736 study in PDL-1i for use in metastatic bladder cancer, showed 46.4% response rate and granted accelerate approval by FDA in May 2017. (2)
AVELUMAB - Received accelerated approval in 2017 based on JAVELIN study.
COMBO THERAPY - CheckMate 032 included nivolumab/ipilimumab
ERDAFITINIB - Post chemotherapy, post immunotherapy, FGF receptor target. NEJM published study in 2019 showed objective tumor response in 40% patients after disease post chemotherapy progression. Provided 5.5mo PFS, 13.8 OS. This study included patients with visceral metastasis and more than half w/ GFR <60. (6)
ENFORTUMAB
SIDE EFFECTS OF IMMUNOTHERAPY
Immune mediated adverse events (IMAE's) are activively monitored. Any organ system can be affected and neuropathies and arthralgias can also occure. The most commonly experienced one may include pneumoitis., colitis, dermatitis, hepatic transaminitis and thyroiditis. (10)
- Bellmunt, Joaquim, et al. "Pembrolizumab as second-line therapy for advanced urothelial carcinoma." New England Journal of Medicine 376.11 (2017): 1015-1026.
- Massard, Christophe, et al. "Safety and efficacy of durvalumab (MEDI4736), an anti-programmed cell death ligand-1 immune checkpoint inhibitor, in patients with advanced urothelial bladder cancer." Journal of clinical oncology: official journal of the American Society of Clinical Oncology 34.26 (2016): 3119-3125.
- Shepherd, Andrew RH, Emily Shepherd, and Nicholas R. Brook. "Intravesical Bacillus Calmette-Guerin with interferon-alpha versus intravesical Bacillus Calmette-Guerin for treating non-muscle-invasive bladder cancer." status and date: New, published in 3 (2016).
- Vaughn, David J., et al. "Health-Related Quality-of-Life Analysis From KEYNOTE-045: A Phase III Study of Pembrolizumab Versus Chemotherapy for Previously Treated Advanced Urothelial Cancer." Journal of Clinical Oncology(2018): JCO-2017.
- US FDA. FDA Alerts Health Care Professionals and Oncology Clinical Investigators About and Efficacy Issue Identified in Clinical Trials or Some Patients Taking Keytruda (pemnrolizumab) ot Tecentriq (atezolizumab) as Monotherapy to Treat Urothelial Cancer With Low Expression of PD-La. Updated August 16, 2018. Acessed September 30, 2018.
- https://www.nejm.org/doi/full/10.1056/NEJMoa1817323
- Powles, T. B., Durán, I., Heijden, M. S. van der, Loriot, Y., Vogelzang, N. J., Giorgi, U. D., … Bamias, A. (2017). Atezolizumab versus chemotherapy in patients with platinum-treated locally advanced or metastatic urothelial carcinoma (IMvigor211): a multicentre, open-label, phase 3 randomised controlled trial. The Lancet, 391(10122), 748–757.
- https://www.urotoday.com/journal/everyday-urology-oncology-insights/articles/113271-immune-oncology-the-urologist-s-role.html
- Castellano D. ESMO 2018: ABACUS, A phase II Study Investigating the Safety and Efficacy of Neoadjuvant Atezolizumab in Muscle Invasive Bladder Cancer. ESMO 2018 Congress. Munich, Germany2018.
- https://www.urotoday.com/journal/everyday-urology-oncology-insights/articles/120672-the-advanced-bladder-clinic-are-you-ready.html. Accessed 10/17/2020
- Hopkins, Ashley M., et al. "Concomitant Proton Pump Inhibitor Use and Survival in Urothelial Carcinoma Treated with Atezolizumab." Clinical Cancer Research (2020).
- Necchi, Andrea, et al. "Pembrolizumab as neoadjuvant therapy before radical cystectomy in patients with muscle-invasive urothelial bladder carcinoma (PURE-01): an open-label, single-arm, phase II study." Journal of Clinical Oncology 36.34 (2018): 3353-3360.
- Bajorin, Dean F., et al. "Adjuvant nivolumab versus placebo in muscle-invasive urothelial carcinoma." New England Journal of Medicine 384.22 (2021): 2102-2114.