Renal Cell Carcinoma Studies
- RANDOMIZED TRIALS FOR ADJUVANT IMMUNOTHERAPY for ADVANCED RCC
- Combo better for intermediate and poor risk patients (IMDC stratification ) but Sunitinib better for good risk
- Nivolumab (PD1i), Ipilimumab (CTLA4i)
- 18mo OS 75% w/ NI vs. 60% w/ Sunitinib alone
- 9% complete response w/ NI vs. 1% w/ Sunitinib
- Median OS not reached yet w/ NI, 26.0 mo w/ Sunitinib
- High side effect rate for both: grade 3 or 4 events 46% in NI, 63% in Sunitinib
- 22% discontinued in NI vs. 12% in Sunitinib
- 60% of combo required IV corticosteroids for adverse effects
- PFS better in PDL1+ but not PDL1- paatients. OS better in both groups, moreso in PDL1+
- On April 16, 2018 FDA approved nivolumab (Opdivo) and ipilimumab (Yervoy) in combination for the treatment of intermediate- or poor-risk, previously untreated advanced renal cell carcinoma (RCC).
INMOTION151 - 1st line Bevacizumab/atezolizumab vs. sunitinib in treatment naive advanced/metastatic RCC (10)
- Allowed sarcomatoid histology in addition to clear cell
- PFS better with combo in PDL1+
- CR 9% in combo vs. 4% in sunitinib
- 16% of combo group required corticosteroids
- Less adverse effects than sunitinib
- OS data immature
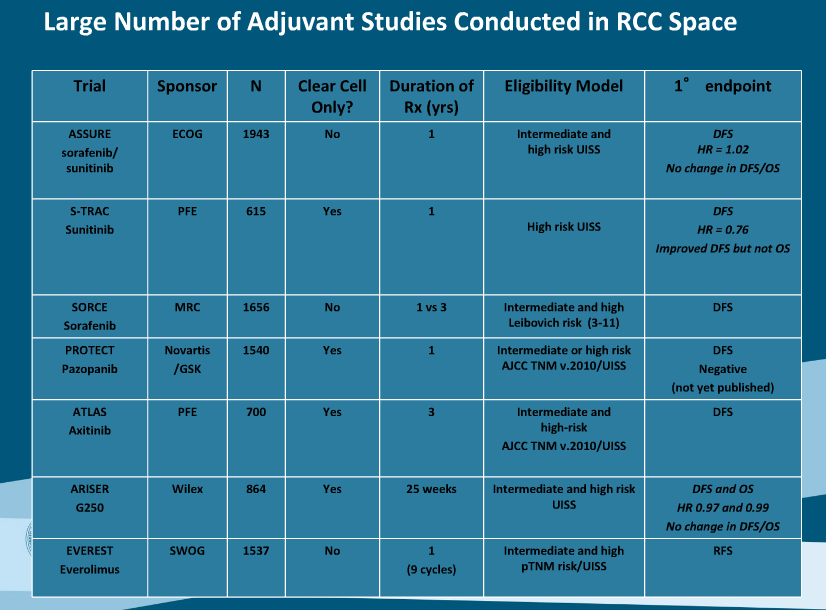
S-TRAC (Sunitinib) (615 patients) - 1 yr sunitinib improved recurrence-free survival by 1.2 years in high risk local recurrence (2) and in a subgroup of high risk patients by 2.2 years. No OS benefit at 5 years follow-up
- Included more high grade disease, pT3 or greater: more than 1/3 of patients with high grade T1 or T2 disease that were included in ASSURE would not have been included in S-TRAC
- Only clear cell histology
- Doses were reduced by 12.5mg but only if required, resulting in 1/3 patients with dose reduction
- 45% drop out rate due to toxicity
- More frequent imaging than ASSURE
- Independent non-blinded central review (2)
Sunitinib vs. IFN -
- 750patients w/ treatment naive mRCC
- Median overall survival was greater in the sunitinib group than in the IFN-alpha group (26.4 v 21.8 months (9)
RANDOMIZED TRIALS FOR ADJUVANT IMMUNOTHERAPY in NON-METASTATIC RCC
ASSURE (Sunitinib or Sorafenib) (1943 patients) - no recurrence-free survival benefit over placebo in adjuvant therapy (1)
- Included patients w/ pT1b or greater grade 3 to 4 disease
- Median DFS of the placebo group 1 year longer in ASSURE than S-TRAC, reflecting the worse disease grade in S-TRAC
- 20% had non-clear cell histology, which is not driven by aberrant VHL/HIF1alpha pathway; however further subroup analysis of clear cell only histology failed to shows survival benefit
- Doses were decreased from 50mg to 37.5mg in all patients in 2009 and increased as tolerated
- 44% drop out rate due to toxicity
- Blinded investigator assessed end-points
CYTOREDUCTIVE NEPHRECTOMY
CARMENA - Nephrectomy + Sunitinib (13.9mo OS) is inferior to Sunitinib alone (18.4mo OS) in intermediate and poor risk patient with mRCC. (8)
RANDOMIZED TRIALS FOR LYMPHADENECTOMY
EORTC 30881 (5)
- no survival benefit for clinically negative nodes
- however, 77% of patients were low grade with 4% LN involvement
- this population likely doesn't represent those who would likely benefit
OTHERS
SORCE (Sorafenib in Treating Patients at Risk of Relapse after Undergoing Surgery to Remove Kidney Cancer, NCT00492258) - results pending
ATLAS (Adjuvant Axitinib Therapy of Renal Cell Cancer in High Risk Patients, NCT01599754) - clear cell T3 or T4 - results pending
EVEREST (Everolimus in Treating Patients with Kidney Cancer Who Have Undergone Surgery, NCT01120249) clear or papillary w/ high grade T1b or T2 to T4 tumors to everolimus or placebo for 1 year
PROTECT (A study to evaluate Pazopanib as an Adjuvant Treatment for Localized RCC, NCT01235962)
CHECKMATE025 (Nivolumab vs. Everolimus in pretreated advanced or metastatic clear cell RCC, NCT01668784) (3)
- OS 5.4mo longer w/ nivolumab
- Nivolumab has significantly lower number of side effects and improved quality of life (4)
INMOTION010 (adjuvant Atezolizumab in patients w/ high risk of developing metastasis, NCT03024996)
PT2385 is the first medication from a new class of drugs called hypoxia-inducible factor-2α (HIF-2α) antagonists.
- Complete response, partial response, and stable disease as best response were achieved by 2%, 12%, and 52% of patients, respectively.
- Most common side effects of anemia (grade 1 to 2, 35%; grade 3, 10%), peripheral edema (grade 1 to 2, 37%; grade 3, 2%), and fatigue (grade 1 to 2, 37%; no grade 3 or 4) (6)
- Haas, Naomi B., et al. "Adjuvant sunitinib or sorafenib for high-risk, non-metastatic renal-cell carcinoma (ECOG-ACRIN E2805): a double-blind, placebo-controlled, randomised, phase 3 trial." The Lancet 387.10032 (2016): 2008-2016.
- Ravaud, Alain, et al. "Adjuvant sunitinib in high-risk renal-cell carcinoma after nephrectomy." New England Journal of Medicine 375.23 (2016): 2246-2254.
- Motzer, Robert J., et al. "Nivolumab versus everolimus in advanced renal-cell carcinoma." New England Journal of Medicine 373.19 (2015): 1803-1813.
- Cella, David, et al. "Quality of life in patients with advanced renal cell carcinoma given nivolumab versus everolimus in CheckMate 025: a randomised, open-label, phase 3 trial." The Lancet Oncology 17.7 (2016): 994-1003.
- Blom, Jan HM, et al. "Radical nephrectomy with and without lymph-node dissection: final results of European Organization for Research and Treatment of Cancer (EORTC) randomized phase 3 trial 30881." European urology 55.1 (2009): 28-34.
- Courtney, Kevin D., et al. "Phase I Dose-Escalation Trial of PT2385, a First-in-Class Hypoxia-Inducible Factor-2α Antagonist in Patients With Previously Treated Advanced Clear Cell Renal Cell Carcinoma." Journal of Clinical Oncology (2017): JCO-2017.
- Motzer, Robert J., et al. "Nivolumab plus Ipilimumab versus Sunitinib in Advanced Renal-Cell Carcinoma." New England Journal of Medicine (2018).
- Méjean, Arnaud, et al. "Sunitinib alone or after nephrectomy in metastatic renal-cell carcinoma." New England Journal of Medicine (2018).
- Motzer, Robert J., et al. "Overall survival and updated results for sunitinib compared with interferon alfa in patients with metastatic renal cell carcinoma." Journal of clinical oncology27.22 (2009): 3584.
- Motzer, Robert J., et al. "IMmotion151: a randomized phase III study of atezolizumab plus bevacizumab vs sunitinib in untreated metastatic renal cell carcinoma (mRCC)." (2018): 578-578.