Urine Markers
There are more than 30 published biomarkers. Cytology has highest specificity, other markers have higher sensitivity.
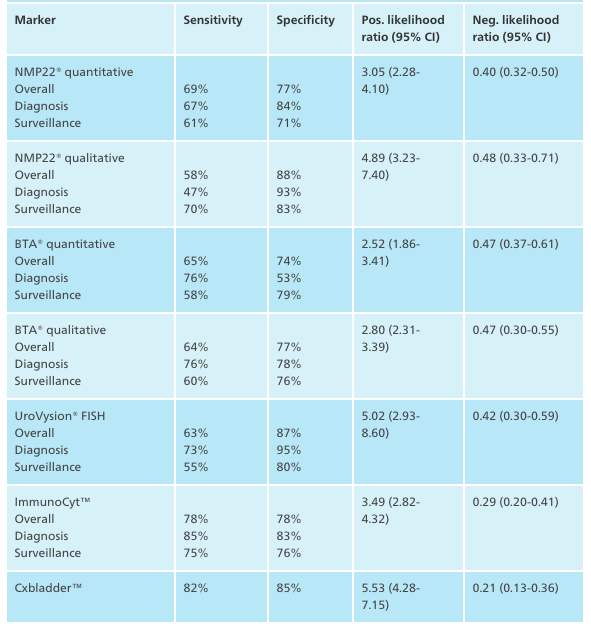
Urovysion FISH
NMP22 (3)
BTA (qualititative=BTA Stat; quantitative=BTA TRAK) (3,4)
GeneXpert Bladder Cancer ASSAY (6)
Cytology (5)
ImmunCyt/uCyt+
AssureMDx
Cxbladder (10)
BladderChek
UROBEST
mRNA's overexpressed in bladder cancer are ABL1, CRH, IFG2, ANXA10, KRT20, AR, PIK3CA, UPK1b, UPK2, MGEA5. (6) IGF-2 urine levels have been shown to be high in patient w/ bladder cancer. (7) GeneXpert uses mRNA testing capability.
URINE METABOLIC BASED BIOMARKERS
Metabolic changes occur that are reflected in urine and early studies into these biomarkers have suggested a high sensitivity and specificity. (9)
Urovysion FISH
- Detects increased copies of chromosome 3, 7, 17 and deletion of 9p21 (site of p16) (1)
- Sensitivity of 58%, 77%, 96%, 100% in grades 1, 2, 3 and CIS respectively (higher than cytology in all grades) (2)
- FDA approved
- Positive FISH after BCG was found to be 3.3 risk of progression/recurrence in one study. (11)
NMP22 (3)
- Available as POC or send out ELISA
- Detects NMP22, a nuclear mitotic protein released from apoptotic cells
- Sensitivity ranged of 67% (from 68.5% to 88.5%) and specificity of 75% (ranges from 65.2% to 91.3%)
- False positives from hematuria, infection, stones, recent instrument
- FDA approved
BTA (qualititative=BTA Stat; quantitative=BTA TRAK) (3,4)
- Available as POC or send out ELISA
- Detects human complement factor H related protein in urine
- Sensitivity reported ranges of 62-68% while specificity reported around 73.6%
- False positives from hematuria, infection, prior BCG
- FDA approved
GeneXpert Bladder Cancer ASSAY (6)
- A 90 minute test
- mRNA's ABL1, CRH, IGF2, ANXA10, UPK1B
- Overall sensitivity was 73% and specificity 90% if hematuria and 77% in surveillance population
- 83% sensitivity for high grade bladder cancer
Cytology (5)
- Sensitivity 35%, specificity 94% overall
- 58% up to 80% sensitivity for intermediate and high grade (5,8)
- approximately 20% in low risk (8)
ImmunCyt/uCyt+
- Looks at 3 monoclonal antibodies associated w/ UCC
- 81% sensitivity, 75% specificity
- False positives from stones, infection, BPH
- Does not come in POC, requires 500cells to make diagnosis
- FDA approved
AssureMDx
- DNA methylation of 3 genes
- 93% sensitivity, 83% specificity, NPV of 99.6%
Cxbladder (10)
- Gene expression of 5 biomarkers
- 93% sensitivity, 85% specificity
- Includes biomarker for inflammation to reduce false positives
BladderChek
UROBEST
mRNA's overexpressed in bladder cancer are ABL1, CRH, IFG2, ANXA10, KRT20, AR, PIK3CA, UPK1b, UPK2, MGEA5. (6) IGF-2 urine levels have been shown to be high in patient w/ bladder cancer. (7) GeneXpert uses mRNA testing capability.
URINE METABOLIC BASED BIOMARKERS
Metabolic changes occur that are reflected in urine and early studies into these biomarkers have suggested a high sensitivity and specificity. (9)
- Schroeder, Grethchen L., et al. "A side by side comparison of cytology and biomarkers for bladder cancer detection." The Journal of urology 172.3 (2004): 1123-1126.
- Grossman, H. Barton, et al. "Detection of bladder cancer using a point-of-care proteomic assay." Jama 293.7 (2005): 810-816.
- Friedrich, M. G., et al. "Comparison of multitarget fluorescence in situ hybridization in urine with other noninvasive tests for detecting bladder cancer." BJU international 92.9 (2003): 911-914.
- Yoo, Jong-Ha, et al. "Analysis of fluorescence in situ hybridization, mtDNA quantification, and mtDNA sequence for the detection of early bladder cancer." Cancer Genetics 198.2 (2010): 107-117.
- Van Rhijn, Bas WG, Henk G. van Der Poel, and Theo H. van Der Kwast. "Urine markers for bladder cancer surveillance: a systematic review." European urology 47.6 (2005): 736-748.
- Wallace, Ellen, et al. "Development of a 90-minute integrated noninvasive urinary assay for bladder cancer detection." The Journal of urology 199.3 (2018): 655-662.
- Watson, Jenny A., et al. "Urinary insulin‐like growth factor 2 identifies the presence of urothelial carcinoma of the bladder." BJU international 103.5 (2009): 694-697.
- Diagnosis and Treatment of Non-Muscle Invasive Bladder Cancer: AUA/SUO Joint Guideline
- Loras, A., et al. "Bladder cancer recurrence surveillance by urine metabolomics analysis." Scientific reports 8.1 (2018): 9172.
- Kavalieris, Laimonis, et al. “Performance Characteristics of a Multigene Urine Biomarker Test for Monitoring for Recurrent Urothelial Carcinoma in a Multicenter Study.” The Journal of Urology, vol. 197, no. 6, 2017, pp. 1419–1426.
- Lotan, Yair, et al. "Evaluation of the fluorescence in situ hybridization test to predict recurrence and/or progression of disease after Bacillus Calmette-Guerin for primary high grade nonmuscle invasive bladder cancer: results from a prospective multicenter trial." The Journal of urology 202.5 (2019): 920-926.