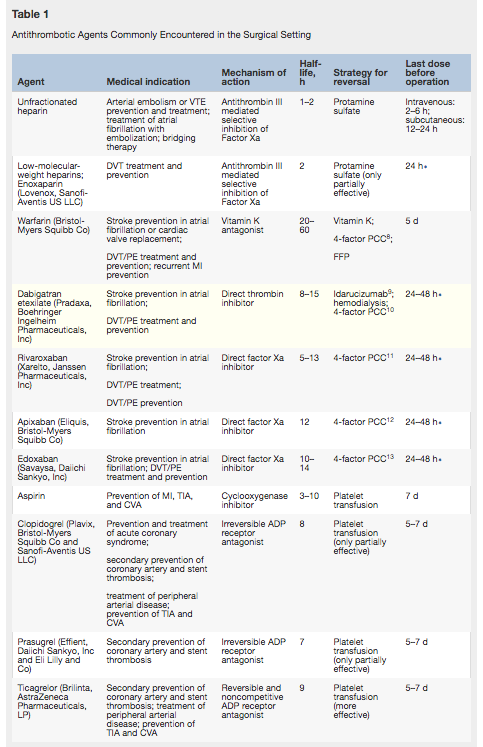
Anticoagulation
The ACS has provided nice guidelines for peri-operative management of anticoagulation (11).
Tinazparin is considered safer if CrCl <20ml/sec (13-16)
COUMADIN
Major bleeding has a case fatality rate of 11% with coumadin. Reverse with 5-10mg vit K and 4F-PCC. (6)
NSAIDS
NOACs
Major bleeding has a case fatality rate of 7.5% with non-vitamin K oral antagonists (NOACs) which have minimal to no anticoagulant effect remaining when therapy with a NOAC is withheld for 48 to 72 hours before surgery with normal kidney function. NOACs have short half-lives, with approximately 80% of their anticoagulant effect gone at 24 hours after ingestion of the last dose when kidney function is preserved. Peak plasma levels of NOACs are reached by approximately 3 hours after intake. (1) For apixaban, edoxaban, rivaroxaban, negative anti-factor Xa assay usually indicated insignificant drug levels. Normal PT/aPTT does not exclude signficant drug levels, wheras prolonged PT suggests clinically significant drug levels. IV 4F-PCC or IV aPCC is the reversal agent to be used (50 IU/KG?). (6)
NOACs REVERSAL
Apixaban (Eliquis) and rivaroxaban (Xarelto) also have a reversal agent (inactivated-zhzo [andexanet alfa; Andexxa - Portola]) which was FDA approved in May 2018. Andexxa is a genetically modified variant of human factor Xa made in the Chinese hamster ovary cell line that binds to Xa inhibitors of all direct (apixaban, betrixaban, edoxaban, and rivaroxaban) and indirect (enoxaparin and fondaparinux) Xa inhibitors. (7) The studies of healthy patients thusfar have used 400mg or 800mg IV bolus +/- 4-8mg/min infusion for 2hrs and results in >90% of recipients within 2-5 minutes having anti-factor Xa activity reduced and thrombin generation fully restored during the continuous infusion. No VTE events occurred in these healthy subjects. (10) A different study of 238 patients with acute major bleeding (61% cerebral, 27% GI) showed 83% of patients had bleeding resolved within 12hrs of taking the reversal agent. Notably 11% have VTE and 12% died within 30 days of administration. (8,9)
Dabigatran (Pradaxa) has a reversal agent (Iadarucizumab [Praxbind]) FDA approved in 2015. (1) Idarucizumab is an IV monoclonal antibody given in 2 boluses (5-15 minutes apart), with total reversal of dabigatran’s anticoagulant effect. 5m of IV normalize dilute TT and ECT in 88-98%. (5) Lab tests for Dabigatran that when normal indicate insignificant drug levels include dilute thrombin time (TT), ecarin clotting time (ECT), ecarin chromogenic assay (ECA). aPTT at normal level usually indicates the drug levels are insignificant and prolonged aPTT suggests significant drug levels. Four factor prothrombin complex concentrate (4F-PCC)/activated prothrombin complex concentrae (aPCC) can also be used to reverse Dabigatran. (6) A TEG study can show the anticoagulant activity of NOACs. (2)
Betrixaban, has been approved by the FDA for VTE prevention among adults who are hospitalized because of acute medical illnesses.
More accurate tests to measure plasma dabigatran include the diluted thrombin time assay, the ecarin clotting time assay, or the ecarin thromboelastometry assay. (3,4)
REVERSAL: (6)
Dabigatran - idarucizumab or IV Four factor prothrombin complex concentrate (4F-PCC)/activated prothrombin complex concentrae (aPCC) (6)
Warfarin- 5-10mg vit K and 4F-PCC (6)
Apixaban, edoxaban, rivaroxaban - Andexxa (Low dose costs $24,250, high dose costs $49,500)
Ticagrelor - Monoclonal antibody reversal - reversal occurred within 5 minutes after the initiation of PB2452 and was sustained for more than 20 hours (12)
LAB TESTS (6)
Dabigitran - normal indicate insignificant drug levels include dilute thrombin time (TT), ecarin clotting time (ECT), ecarin chromogenic assay (ECA). aPTT at normal level usually indicates the drug levels are insignificant and prolonged aPTT suggests significant drug levels. (6)
Apixaban, edoxaban, rivaroxaban - Negative anti-factor Xa assay usually indicated insignificant drug levels. Normal PT/aPTT does not exclude signficant drug levels, wheras prolonged PT suggests clinically significant drug levels.
This is a good basic outline and review of TEG. (17)
Tinazparin is considered safer if CrCl <20ml/sec (13-16)
COUMADIN
Major bleeding has a case fatality rate of 11% with coumadin. Reverse with 5-10mg vit K and 4F-PCC. (6)
NSAIDS
NOACs
Major bleeding has a case fatality rate of 7.5% with non-vitamin K oral antagonists (NOACs) which have minimal to no anticoagulant effect remaining when therapy with a NOAC is withheld for 48 to 72 hours before surgery with normal kidney function. NOACs have short half-lives, with approximately 80% of their anticoagulant effect gone at 24 hours after ingestion of the last dose when kidney function is preserved. Peak plasma levels of NOACs are reached by approximately 3 hours after intake. (1) For apixaban, edoxaban, rivaroxaban, negative anti-factor Xa assay usually indicated insignificant drug levels. Normal PT/aPTT does not exclude signficant drug levels, wheras prolonged PT suggests clinically significant drug levels. IV 4F-PCC or IV aPCC is the reversal agent to be used (50 IU/KG?). (6)
NOACs REVERSAL
Apixaban (Eliquis) and rivaroxaban (Xarelto) also have a reversal agent (inactivated-zhzo [andexanet alfa; Andexxa - Portola]) which was FDA approved in May 2018. Andexxa is a genetically modified variant of human factor Xa made in the Chinese hamster ovary cell line that binds to Xa inhibitors of all direct (apixaban, betrixaban, edoxaban, and rivaroxaban) and indirect (enoxaparin and fondaparinux) Xa inhibitors. (7) The studies of healthy patients thusfar have used 400mg or 800mg IV bolus +/- 4-8mg/min infusion for 2hrs and results in >90% of recipients within 2-5 minutes having anti-factor Xa activity reduced and thrombin generation fully restored during the continuous infusion. No VTE events occurred in these healthy subjects. (10) A different study of 238 patients with acute major bleeding (61% cerebral, 27% GI) showed 83% of patients had bleeding resolved within 12hrs of taking the reversal agent. Notably 11% have VTE and 12% died within 30 days of administration. (8,9)
Dabigatran (Pradaxa) has a reversal agent (Iadarucizumab [Praxbind]) FDA approved in 2015. (1) Idarucizumab is an IV monoclonal antibody given in 2 boluses (5-15 minutes apart), with total reversal of dabigatran’s anticoagulant effect. 5m of IV normalize dilute TT and ECT in 88-98%. (5) Lab tests for Dabigatran that when normal indicate insignificant drug levels include dilute thrombin time (TT), ecarin clotting time (ECT), ecarin chromogenic assay (ECA). aPTT at normal level usually indicates the drug levels are insignificant and prolonged aPTT suggests significant drug levels. Four factor prothrombin complex concentrate (4F-PCC)/activated prothrombin complex concentrae (aPCC) can also be used to reverse Dabigatran. (6) A TEG study can show the anticoagulant activity of NOACs. (2)
Betrixaban, has been approved by the FDA for VTE prevention among adults who are hospitalized because of acute medical illnesses.
More accurate tests to measure plasma dabigatran include the diluted thrombin time assay, the ecarin clotting time assay, or the ecarin thromboelastometry assay. (3,4)
REVERSAL: (6)
Dabigatran - idarucizumab or IV Four factor prothrombin complex concentrate (4F-PCC)/activated prothrombin complex concentrae (aPCC) (6)
Warfarin- 5-10mg vit K and 4F-PCC (6)
Apixaban, edoxaban, rivaroxaban - Andexxa (Low dose costs $24,250, high dose costs $49,500)
- if ≤10mg Xarelto of ≤5mg Eliquis get low dose regimen: 400mg IV bolus and 4mg/min for 2hrs
- if >10mg Xarelto or >5mg Eliquis w/ last dose <8hrs get high dose regimen: 800mg IV bolus and 8mg/min for 2 hrs. If last dose ≥8hrs get low dose
Ticagrelor - Monoclonal antibody reversal - reversal occurred within 5 minutes after the initiation of PB2452 and was sustained for more than 20 hours (12)
LAB TESTS (6)
Dabigitran - normal indicate insignificant drug levels include dilute thrombin time (TT), ecarin clotting time (ECT), ecarin chromogenic assay (ECA). aPTT at normal level usually indicates the drug levels are insignificant and prolonged aPTT suggests significant drug levels. (6)
Apixaban, edoxaban, rivaroxaban - Negative anti-factor Xa assay usually indicated insignificant drug levels. Normal PT/aPTT does not exclude signficant drug levels, wheras prolonged PT suggests clinically significant drug levels.
This is a good basic outline and review of TEG. (17)
- Verma, Atul, et al. "What Surgeons Should Know About Non–Vitamin K Oral Anticoagulants: A Review." JAMA Surgery(2018).
- Dias, João D., et al. "Use of thromboelastography (TEG) for detection of new oral anticoagulants." Archives of Pathology and Laboratory Medicine 139.5 (2015): 665-673.
- Božič-Mijovski, Mojca, et al. "Diluted thrombin time reliably measures low to intermediate plasma dabigatran concentrations." Annals of clinical biochemistry 53.4 (2016): 446-451.
- Körber, Mareike Kristina, et al. "In vitro and ex vivo measurement of prophylactic dabigatran concentrations with a new ecarin-based thromboelastometry test." Transfusion Medicine and Hemotherapy 44.2 (2017): 100-105.
- Pollack Jr, Charles V., et al. "Idarucizumab for dabigatran reversal." New England Journal of Medicine 373.6 (2015): 511-520.
- Anderson, Irsk, and Adam S. Cifu. "Management of Bleeding in Patients Taking Oral Anticoagulants." Jama 319.19 (2018): 2032-2033.
- Rogers, Kelly C., and Shannon W. Finks. "A New Option for Reversing the Anticoagulant Effect of Factor Xa Inhibitors: Andexanet alfa (Andexxa®)." The American journal of medicine (2018).
- Connolly, Stuart J., et al. "Andexanet alfa for acute major bleeding associated with factor Xa inhibitors." New England Journal of Medicine 375.12 (2016): 1131-1141.
- Connolly, Stuart J. ANNEXA-4 suggests experimental drug controls bleeding in patients taking factor Xa inhibitors. Presented at: ACC.18; March 10-12, 2018; Orlando FL.
- Siegal, Deborah M., et al. "Andexanet alfa for the reversal of factor Xa inhibitor activity." New England Journal of Medicine373.25 (2015): 2413-2424.
- Hornor, Melissa A., et al. "American College of Surgeons' Guidelines for the Perioperative Management of Antithrombotic Medication." Journal of the American College of Surgeons 227.5 (2018): 521-536.
- Bhatt, D. L., Pollack, C. V., Weitz, J. I., Jennings, L. K., Xu, S., Arnold, S. E., … Lee, J. S. (2019). Antibody-Based Ticagrelor Reversal Agent in Healthy Volunteers. The New England Journal of Medicine.
- Shea-Budgell, M. A., et al. “Evidence-Based Guidance on Venous Thromboembolism in Patients with Solid Tumours.” Current Oncology, vol. 21, no. 3, 2014, pp. 504–514.
- Nicolaides, AN, et al. “Prevention and Treatment of Venous Thromboembolism - International Consensus Statement (Guidelines According to Scientific Evidence).” International Angiology, vol. 25, no. 2, 2006, pp. 101–161.
- Mahé, Isabelle, et al. “Tinzaparin and Enoxaparin given at Prophylactic Dose for Eight Days in Medical Elderly Patients with Impaired Renal Function - A Comparative Pharmacokinetic Study.” Thrombosis and Haemostasis, vol. 97, no. 4, 2007, pp. 581–586.
- Siguret, V., et al. “No Accumulation of the Peak Anti-Factor Xa Activity of Tinzaparin in Elderly Patients with Moderate-to-Severe Renal Impairment: The IRIS Substudy.” Journal of Thrombosis and Haemostasis, vol. 9, no. 10, 2011, pp. 1966–1972.
- Subramanian, Madhu, Lewis J. Kaplan, and Jeremy W. Cannon. "Thromboelastography-Guided Resuscitation of the Trauma Patient." JAMA surgery 154.12 (2019): 1152-1153.