PEMBROLIZUMAB
Programmed cell death protein 1 (PD-1) antibody
METASTATIC BLADDER CANCER:
Phase Ib KEYNOTE-012 (1)
Phase II KEYNOTE-052 (2)
Phase III KEYNOTE-045 (3) = Pembro vs. chemotherapy
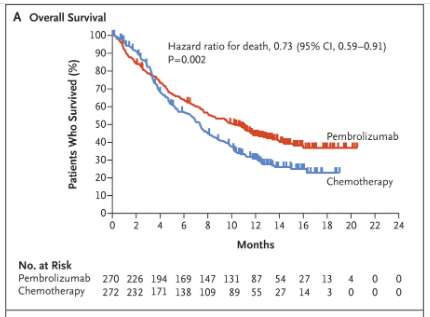
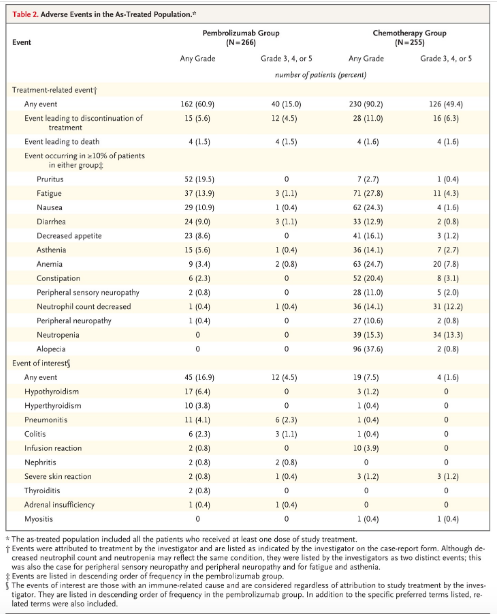
Keynote 045 showed pembrolizumab was associated with significantly longer overall survival (by approximately 3 months) and with a lower rate of treatment-related adverse events than chemotherapy as second-line therapy for platinum-refractory advanced urothelial carcinoma. Showed 2.1mo PFS, 10.3mo OS, 44% 12mo survival (table 1 below). (3) KEYNOTE-045 found pembrolizumab offered stable or improved measures of global health status and quality of life compared with chemotherapy in a phase III trial of patients with previously treated advanced urothelial cancer. (7) The FDA issued a May 2018 safety alert that people with low PD-L1 expression have decreased survival with pembrolizumab or atezolizumab compared to cisplatin or carboplatin based therapy. In August they added the requirement for a diagnostic test be done in anyone on these drugs to test for PD-L1 expression. (8)
METASTATIC BLADDER CANCER:
Phase Ib KEYNOTE-012 (1)
Phase II KEYNOTE-052 (2)
Phase III KEYNOTE-045 (3) = Pembro vs. chemotherapy
Keynote 045 showed pembrolizumab was associated with significantly longer overall survival (by approximately 3 months) and with a lower rate of treatment-related adverse events than chemotherapy as second-line therapy for platinum-refractory advanced urothelial carcinoma. Showed 2.1mo PFS, 10.3mo OS, 44% 12mo survival (table 1 below). (3) KEYNOTE-045 found pembrolizumab offered stable or improved measures of global health status and quality of life compared with chemotherapy in a phase III trial of patients with previously treated advanced urothelial cancer. (7) The FDA issued a May 2018 safety alert that people with low PD-L1 expression have decreased survival with pembrolizumab or atezolizumab compared to cisplatin or carboplatin based therapy. In August they added the requirement for a diagnostic test be done in anyone on these drugs to test for PD-L1 expression. (8)
NONMETASTATIC BLADDER CANCER
Phase II KEYNOTE-057 (4)
2020 NCCN guideline includes pembrolizumab for BCG unresponsive high risk non muscle invasive bladder cancer with Tis with or without papillary tumors who are ineligible or elect not to undergo cystectomy. (5)
KEYNOTE-057 - Evaluated pembrolizumab 200mg q3weeks for 24mo or until disease recurrence in 103 cystectomy ineligible or refusing patients with high risk, BCG unresponsive NMIBC with CIS. 3mo CRR rate 38.8%. 18% of patients had adverse events with most common pruritus (10.7%), fatigue (9.7%), diarrhea (8.7%), hypothyroidism (5.8%), and maculopapular rash (5.8%). Grade 3/4 treatment-related AEs occurred in 13 (12.6%) pts; 1 death was considered treatment-related (colitis in patient inadequately treated with steroids). Immune-mediated AEs occurred in 19 (18.4%) pts. (4)
NEOADJUVANT
Phase II PURE-01 (6)
PURE-01 - 114 patietns with T2-T4a MIBC (with 5% N1) had 37% complete pathologic response (T0) with 55% downstaged to T1. Of note, 19% in this trial had variant histology. (6)
Phase II KEYNOTE-057 (4)
2020 NCCN guideline includes pembrolizumab for BCG unresponsive high risk non muscle invasive bladder cancer with Tis with or without papillary tumors who are ineligible or elect not to undergo cystectomy. (5)
KEYNOTE-057 - Evaluated pembrolizumab 200mg q3weeks for 24mo or until disease recurrence in 103 cystectomy ineligible or refusing patients with high risk, BCG unresponsive NMIBC with CIS. 3mo CRR rate 38.8%. 18% of patients had adverse events with most common pruritus (10.7%), fatigue (9.7%), diarrhea (8.7%), hypothyroidism (5.8%), and maculopapular rash (5.8%). Grade 3/4 treatment-related AEs occurred in 13 (12.6%) pts; 1 death was considered treatment-related (colitis in patient inadequately treated with steroids). Immune-mediated AEs occurred in 19 (18.4%) pts. (4)
NEOADJUVANT
Phase II PURE-01 (6)
PURE-01 - 114 patietns with T2-T4a MIBC (with 5% N1) had 37% complete pathologic response (T0) with 55% downstaged to T1. Of note, 19% in this trial had variant histology. (6)
- Plimack, Elizabeth R., et al. "Safety and activity of pembrolizumab in patients with locally advanced or metastatic urothelial cancer (KEYNOTE-012): a non-randomised, open-label, phase 1b study." The Lancet Oncology 18.2 (2017): 212-220.
- Balar, Arjun V., et al. "First-line pembrolizumab in cisplatin-ineligible patients with locally advanced and unresectable or metastatic urothelial cancer (KEYNOTE-052): a multicentre, single-arm, phase 2 study." The Lancet Oncology 18.11 (2017): 1483-1492.
- Bellmunt, Joaquim, et al. "Pembrolizumab as second-line therapy for advanced urothelial carcinoma." New England Journal of Medicine 376.11 (2017): 1015-1026.
- Balar, Arjun Vasant, et al. "Keynote 057: Phase II trial of Pembrolizumab (pembro) for patients (pts) with high-risk (HR) nonmuscle invasive bladder cancer (NMIBC) unresponsive to bacillus calmette-guérin (BCG)." (2019): 350-350.
- https://www.nccn.org/professionals/physician_gls/PDF/bladder.pdf. Accessed 10/17/20
- Necchi, Andrea, Andrea Anichini, Daniele Raggi, Alberto Briganti, Simona Massa, Roberta Lucianò, Maurizio Colecchia et al. "Pembrolizumab as neoadjuvant therapy before radical cystectomy in patients with muscle-invasive urothelial bladder carcinoma (PURE-01): an open-label, single-arm, phase II study." Journal of Clinical Oncology 36, no. 34 (2018): 3353-3360.
- Vaughn, David J., et al. "Health-Related Quality-of-Life Analysis From KEYNOTE-045: A Phase III Study of Pembrolizumab Versus Chemotherapy for Previously Treated Advanced Urothelial Cancer." Journal of Clinical Oncology(2018): JCO-2017.
- US FDA. FDA Alerts Health Care Professionals and Oncology Clinical Investigators About and Efficacy Issue Identified in Clinical Trials or Some Patients Taking Keytruda (pemnrolizumab) ot Tecentriq (atezolizumab) as Monotherapy to Treat Urothelial Cancer With Low Expression of PD-La. Updated August 16, 2018. Acessed September 30, 2018.